There have been numerous studies over the last century showing impressive results in alleviating magnesium deficiency with both magnesium sulfate (commonly called epsom salt) and magnesium chloride to alleviate magnesium deficiency symptoms. But, what’s the difference?
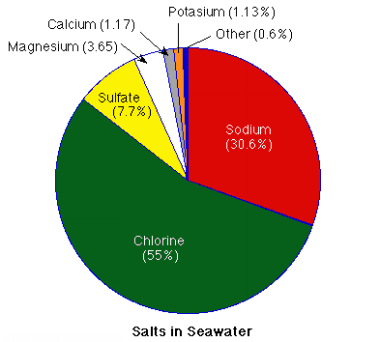
Magnesium chloride and epsom salt (magnesium sulfate, or sulphate) compounds are the most common forms of magnesium found in sea water. Individually, sulfate represents about 7.7%, magnesium approx 3.65% and chlorine 55%.
Elements in Sea Water: There are about 90 elements found in sea water. Each litre contains about 35 g of dissolved salts (3.5%), of which sodium and chlorine make up more than 90%. Together with magnesium, sulfur and calcium, these dissolved salts represent a total of 99%. Add only three more elements – potassium, bromine and carbon – and you have more than 999 parts in a 1000 of the dissolved mass in seawater.

70% of the body’s negative ions (anions) are chloride ions. Interestingly, magnesium chloride is more readily taken up by the body, as chloride makes up such a large amount of electrolyte content. In other words, the body is very hungry for chloride – in particular magnesium chloride, compared to magnesium sulfate.
When you purchase epsom salt (magnesium sulfate) from the supermarket however, it is usually not one harvested from ocean water, but manufactured in a factory as an isolate which is magnesium sulfate. It does not contain the other sea trace minerals that would be present in dehydrated sea salt. Epsom salt (magnesium sulfate) is very cheap and in the event you can’t get hold of anything else, it can certainly save your life.
When you purchase magnesium chloride on the other hand, it is usually harvested from sea water sources that are dehydrated from open ocean, alpine salt pockets, dessert salt pans and lakes, or desalination plants. This salt also has about 2% of other trace minerals still wrapped up in the complex. The other great benefit is that the elemental magnesium content of these bischofite salts ranges between 12 to 16%, depending on salt water source. Laboratory testing of Elektra Magnesium’s magnesium chloride salt flakes shows 15 to 16% elemental magnesium, whilst epsom salt’s elemental magnesium content is only 9 to 10%.
The majority (about 85%) of magnesium chloride flakes in the world are industrial grade, which be used for making magnesium metal alloys, fireworks, or for road de-icing, air cleaning at mining operations, used as soil fertilizer or in aquaculture systems, salt swimming pool chlorination or as bath salts. For ingestion however, you need to look for ‘food grade’, as in Elektra Magnesium Flakes.
FOOD GRADE magnesium chloride is best:
Food grade magnesium chloride flakes are mostly used for making tofu by Asian food manufacturers, for mineralising filtered drinking water or for use as a horse feed supplement. Pharmaceutical grade magnesium chloride is also made by further processing of the food grade flakes and removing all the extra trace minerals.
 The best magnesium chloride source waters for food grade salts are deep ocean or pristine alpine regions not near industry, mining, agriculture or large populations. These sources are the least likely to contain human industrial or sewage waste waters. However, even within these pristine regions magnesium chloride can contain contaminants if harvested using extraction mining techniques. Open salt water lake harvesting offers the best results to get food grade quality. And then further laboratory testing can confirm it.
The best magnesium chloride source waters for food grade salts are deep ocean or pristine alpine regions not near industry, mining, agriculture or large populations. These sources are the least likely to contain human industrial or sewage waste waters. However, even within these pristine regions magnesium chloride can contain contaminants if harvested using extraction mining techniques. Open salt water lake harvesting offers the best results to get food grade quality. And then further laboratory testing can confirm it.
Epsom salt (magnesium sulfate) supplement compared to natural sulfur in foods:
We need sulfate, but in smaller amounts. Epsom salt (magnesium sulfate) is rapidly excreted through the kidneys so that the sulfate doesn’t hang around more than we need it too. This means the magnesium component goes out with it often before enough can be used.
DETOXIFICATION VIA SULFATE: Sulfate exerts an important influence over almost every cellular function. It attaches to phenols and makes them less harmful, and sets them up for being excreted from the kidneys. Sulfate is also used in the blood transport system to carry nutrients in a non-activated state so they don’t coagulate. In this way it helps to keep the blood more fluid, which helps the cardiovascular system and micro-capillary access.
People on the autistic spectrum have compromised detox sulfation pathways (methylation), which means they easily accumulate chemicals and waste products, becoming eventually chemically sensitive and prone to inflammation and ‘clogged tubes’. It means they can do with a bit more sulfate than the average person.
Normally people get plenty of the sulfates they need by eating organic sulfur-containing foods such as garlic, onions, sesame seeds, eggs, fermented foods and bone broths (meat soups). The natural sulfur in these foods is converted by the body to sulfate as is needed, and thereby does not over-supply sulfate. Some people are allergic to ingested sulfates. Natural sulfur-containing foods also tend to be high in the other most important mineral and great co-factor with magnesium – zinc.
People on the spectrum also invariably have digestive issues and gut problems, associated with a low magnesium status – and consequently higher sensitivity to stress. Magnesium is a great natural calming mineral without negative side effects. The best way to calm down a stress or anxiety attack is with a good magnesium salt soak, big magnesium water drink, followed by a magnesium rub with magnesium cream, lotion and/or oil. Epson salt baths or magnesium chloride baths can be very beneficial for detoxification and to promote better sleep, but remember that the magnesium chloride offers a higher concentration of magnesium and is also better for skin care and doesn’t leave skin dry and itchy as is often the case with epson salt baths.
WARNING FOR PREGNANT WOMEN: Epsom salt can be used orally to induce a bowel purge in the case of constipation, but pregnant women should be careful using this strategy because a huge diarrhea event can also trigger a spontaneous abortion.
Magnesium chloride is more hydrating:
An epsom salt bath can be a better alternative to taking it orally, as well as being very relaxing. Bathing in it however can feel harsher on skin compared to magnesium chloride bathing because of a dehydrating effect. Industry uses epsom salt to tan hides! Sulfate is a skin barrier disruptor like alcohol or detergent. This means that over time and with regular epsom salt bathing, the sulfate part will deplete protective skin barrier oils, causing a skin drying effect. Beware also of sulfates in toiletries (like sodium laurel sulfate).
Magnesium chloride hexahydrate on the other hand is hydrating and leaves the skin feeling soft and smooth. Regular magnesium chloride footsoaks prevent build-up of hard dry dead skin layers on heels, which eliminates the need to exfoliate or shave skin off.
‘Hexahydrate’ means the magnesium chloride is complexed with 6 water molecules in a crystal matrix which attracts water molecules.

The flakes are hydrophilic and draw moisture from the air. If left open to the atmosphere long enough the extra water molecules from the air will liquefy the crystals until they become ‘magnesium oil’ (ie. magnesium chloride in water solution). In the right dilution this electrolyte water has the right charge for cell uptake – like a key in a lock.

More optimal uptake because the body is hungry for chloride
70% of the body’s negative ions (anions) are chloride ions. In order to gain access to the intracellular spaces we need potassium and magnesium to be complexed as a chloride to ‘ionise’ them and become completely water soluble. A cytoplasm of structured water makes up the inside of cells, which depends on the presence of enough charge provided by magnesium, potassium and chloride. This plasma is slippery and helps cell components and nutrients to be transported to and fro easily. As the body is exceptionally hungry for the chloride ion as well as the magnesium ion, and as magnesium chloride is fully water-soluble, it will readily take up magnesium chloride in solution. Magnesium also guards the cell membrane channels.
Magnesium chloride hexahydrate comes packaged with other sea trace minerals ‘a-la-Nature’ and also gives you more bang for your buck, as only less than half the volume of magnesium chloride flakes is required in a bath compared to epsom salt for a similar relaxation effect.
Tablets and powders – hard to digest and absorb:
 Magnesium tablet and powder supplements are comprised of different complexes of magnesium such as magnesium oxide, magnesium citrate and various chelated magnesiums (ie. joined to amino acids), which have varying rates of bio-availability. At the lowest end is magnesium oxide with only 4% bio-availability. The better ones still only hover around 40-50% bio-availability. This means that half or more of what you consume is lost in the toilet.
Magnesium tablet and powder supplements are comprised of different complexes of magnesium such as magnesium oxide, magnesium citrate and various chelated magnesiums (ie. joined to amino acids), which have varying rates of bio-availability. At the lowest end is magnesium oxide with only 4% bio-availability. The better ones still only hover around 40-50% bio-availability. This means that half or more of what you consume is lost in the toilet.
Tablets and powders also contain fillers and binders which can sometimes interfere with magnesium uptake. They are more difficult to digest in the stomach, particularly if stomach acid is low. Access across the gut wall, being very sensitive, is also restricted to low concentrations of magnesium such as what would be in mineral water or foods. Higher concentrations result in diarrhea, which means the magnesium is lost too quickly, despite how desperate your tissue cells may be to get more magnesium. In fact, studies have shown that there is a curvi-linear uptake rate, with the higher the concentration of magnesium, the less it gets through the bowel wall. “Fractional absorption rate is high at low Mg load and decreases exponentially with increasing carrier amounts.”
Food and water magnesium sources are better
All the different compounds of oral magnesium tablets and powders must first be digested and dissociated so that the magnesium can be re-combined as the chloride form and made completely water-soluble for intracellular use. Digestion of these magnesium supplements takes several hours, and absorption via the gut wall is minimal.
Natural magnesium chloride hexahydrate however, once in solution, is already in the right form for cells to take up quickly without requiring further digestion. The average elemental magnesium content of spring waters from around the world range from 30mg to 150mg, depending on the source. This range offers the optimum magnesium uptake via the gut wall before a diminishing return on increase of concentration. If you use one pinch of Elektra Magnesium Chloride Flakes (1/2 gram) to remineralise one litre of filtered water you can make a great tasting mineral water which is more hydrating, and actually mimics spring water. That half a gram would contain about 75-80mg of magnesium optimally taken up because it is diluted enough. In other words, the gut wall is designed to take up magnesium optimally from what is naturally contained in foods and water.
 Consuming natural magnesium via foods and waters is very beneficial, but doesn’t satisfy most people’s needs per day, which can go up even more than 1,000mg today if people are under high stress or suffering debilitating diseases. See how much you may need on this page.
Consuming natural magnesium via foods and waters is very beneficial, but doesn’t satisfy most people’s needs per day, which can go up even more than 1,000mg today if people are under high stress or suffering debilitating diseases. See how much you may need on this page.
The skin however can store and cope with higher salt levels compared to the gut wall. Transdermal magnesium products have more success getting access to the interior of the body via the epidermis because the skin is lipophilic (loves fats and lipids). Magnesium Cream, Lotion and Oil can also be used for muscle relaxation, skin care and as a transdermal magnesium supplement with faster results than oral supplements. The plant oils assist the epidermal absorption of magnesium chloride, especially in the case of dry skin. The better condition of the skin the more optimal the magnesium uptake into the epidermis. The epidermis acts in this way as a reservoir for nutrients (ie. electrolytes and essential fatty acids), making them available for absorption by the capillaries of the dermis layer underneath.
Cells are made of electrolyte water – so ‘charge up’!
We are not only a walking water bladder (being mostly made up of water), but we are also made of the salts (electrolytes) of the earth – of particular importance being the master mineral MAGNESIUM. Health decline, stress, pain and inflammation are always accompanied by a deficiency in magnesium and water. Why not replenish with Nature’s most fundamental, essential and powerful nutrition – magnesium chloride and water?
SHOP NOW

Sandy Sanderson © 2018-2020 www.elektramagnesium.com.au

